Resources
About Mercury
What is Mercury
Mercury is a naturally occurring element commonly found in the earth’s crust. On the periodic table, mercury is represented with the symbol, “Hg” derived from its Greek name, hydrargyrum, meaning “liquid silver”. The element exists in three forms with different properties, usage, and toxicity: elemental (or metallic) mercury, inorganic mercury compounds and organic mercury compounds.
Elemental mercury has relatively low boiling point of 356.7°C and high vapor pressure compare to any other metal. It forms amalgam easily with other elements such as gold and silver easily.
Forms of Mercury

Elemental Mercury
Elemental mercury or metallic mercury is a shiny, silver metal that is liquid at room temperature. It is used in some thermometers, fluorescent light bulbs, electrical switches, mining, and other industrial processes. At room temperature, exposed elemental mercury can evaporate to become an invisible and odorless toxic vapor. If it is heated, it vaporizes readily to become a colorless, odorless gas.
This form of mercury is in its free state and has not reacted with other substances. When mercury has reacted with other substances, it forms a compound.

Inorganic Mercury
Inorganic mercury compounds are formed when mercury reacts with other elements, such as sulfur and oxygen, forming inorganic mercury salts. Found naturally in the environment, mining of ore deposits can produce dust containing inorganic mercury to enter the air. Inorganic mercury can also enter water or soil from rocks, factories, or water treatment facilities.
Currently, the use of mercury salts in consumer products have been discontinued, but these compounds are still used in some products, such as cosmetic lightening creams and soaps. It is also used in some industrial processes and other chemical products like insecticides, pesticides and fertilizers.

Organic Mercury
Organic mercury compounds are formed when mercury reacts and combines with carbon. As the element cycles through the environment, land, air, and water, it undergoes complex physical and chemical reactions that can convert elemental or inorganic mercury into an organic mercury compound. For instance, the most common organic mercury compound found in the environment is methylmercury, which is highly toxic.
Fishes and shellfishes bioaccumulate methylmercury from environmental pollution. The chemically transformed methylmercury is absorbed into phytoplankton (microscopic marine algae), which is a diet for zooplankton (tiny aquatic animals), in turns are feasted upon by small fish. These small fish are baits to the bigger fish who even bigger fish then eat, and on and on. Therefore, the larger the size of the fish, the higher level of methylmercury it contains.
Human Exposure to Mercury

Elemental Mercury
Humans are exposed to it when breathing in air containing elemental mercury vapors. Mercury can also be released into the environment from the burning of fuels and wastes containing mercury, and processing of products containing mercury. Exposure to elemental mercury commonly occurs when mercury is released from a product when it breaks or spilt from a container, although exposure towards mercury during occupational settings happen occasionally too. If the exposed mercury is not contained or cleaned up, with its high vapor pressure, it can evaporate into a toxic vapor, which can be a huge concern in poorly ventilated spaces.

Inorganic Mercury
People are exposed to inorganic mercury salts in both occupational and environmental settings. Industries with a higher risk of exposure to mercury and its compounds include mining, electrical equipment manufacturing, chemical manufacturing and other fields in which mercury is used.
Another main exposure to inorganic mercury is through the usage of cosmetics or skincare products tainted with inorganic mercury. Inorganic mercury are adulterated into unethical skincare products to achieve instant fairness and glowing effect. The amount of inorganic mercury in these products can exceed the percentage level.

Organic Mercury
People inherit organic mercury when they eat seafood, such as fish and shellfish exposed to methylmercury. In general, levels of mercury increase with fish size and age, and the mercury levels vary by species (especially predatory) and location. On top of that, if a child-bearing mother consumes fish containing methylmercury, her developing fetus can be exposed to methylmercury, through the placenta.
Additionally, the human can also be exposed to methylmercury from rice diet. Just like seafood, rice grains gather methylmercury from environmental pollution and its source of cultivations. Fortunately, unlike fish, rice does not bioaccumulate. Therefore the methylmercury level is relatively lower than those detected in fish.
Health Effects of Exposures to Mercury
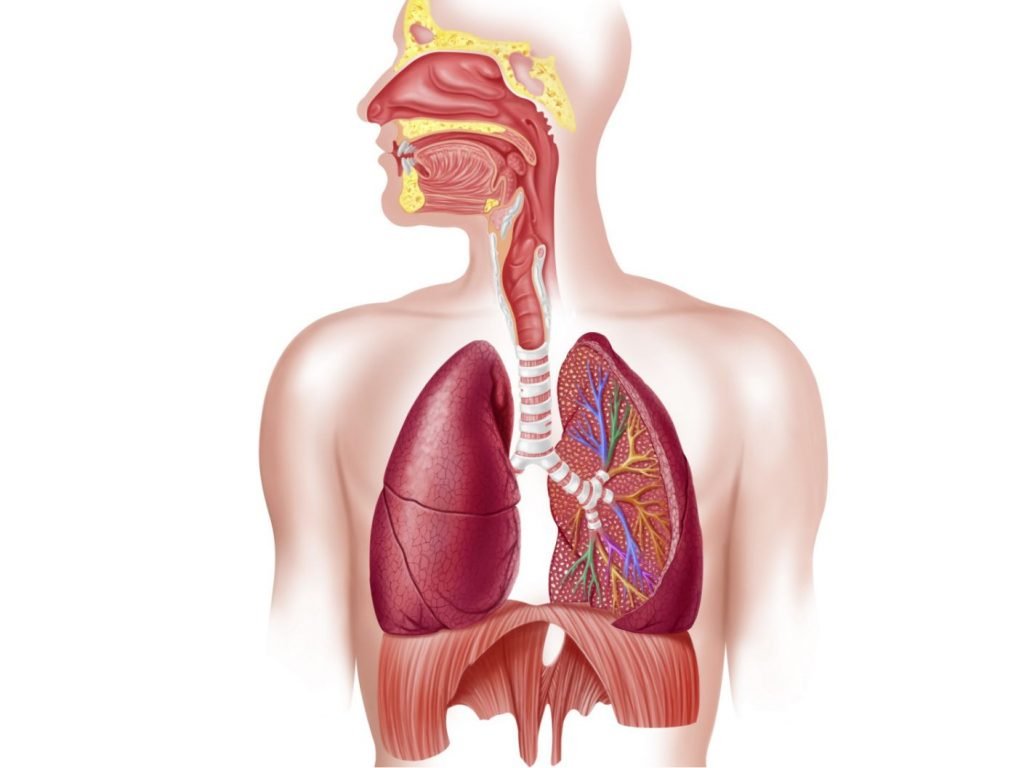
Elemental Mercury
As mentioned, metallic mercury exposed to air can evaporate, becoming an odorless, colorless vapor that is highly toxic. Toxicity occurs when our lungs absorb it as we inhale the toxic vapor. Prolonged exposure at low concentrations may cause mood changes, insomnia, headaches, change in neuromuscular function and neurological disorders. Exposure at high concentrations can cause lung damage resulting in respiratory failure, kidney failure and death.
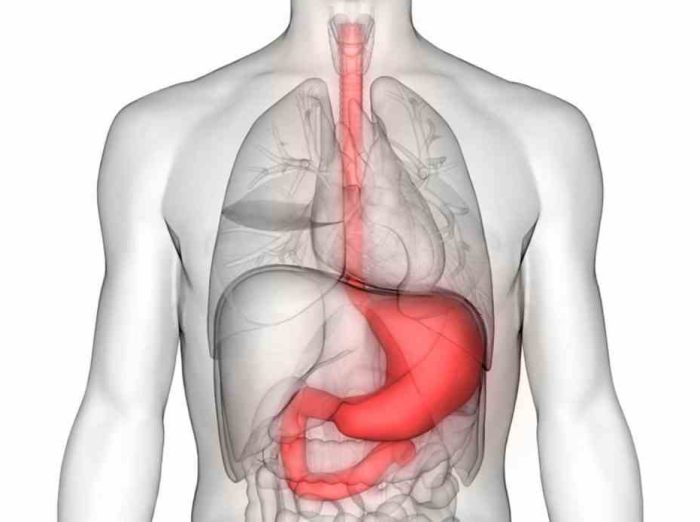
Inorganic Mercury
If consumed in high amounts, the inorganic mercury compounds can irritate and affect the gastrointestinal tract and the digestive system. Repeated applications on the skin can cause dermal absorption and health effects similar to long-term exposure to toxic mercury vapors, which eventually causes organs failure.
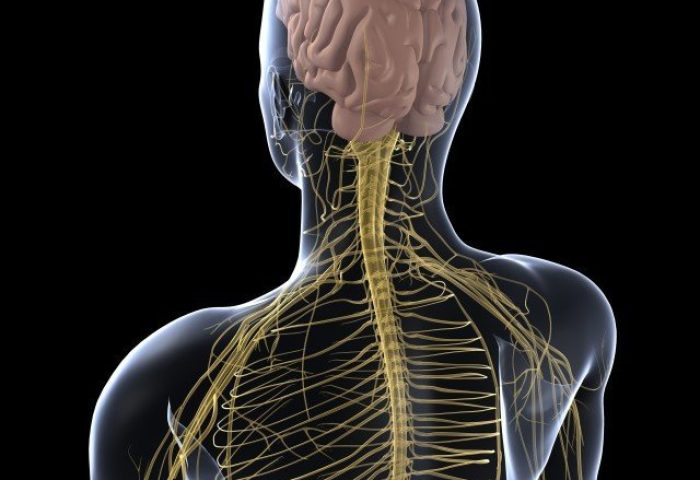
Organic Mercury
Methylmercury exposure commonly occurs when people consume fish and shellfish containing high levels of methylmercury. Prolonged consumption of high amounts of methylmercury can damage the nervous system, leading to symptoms such as muscle weakness, distorted speech, poor muscle coordination and the sensation of “pins and needles” at hands, feet or mouth. On the other hand, during the incidents of Minamata disease, infants exposed to methylmercury from their mother may have developmental disorders and cerebral palsy, causing mercury poisoning in these infants before they were born. This has led to the development of tragic Fetal Minamata Disease.

