Industries & Application
Energy & Chemical
Mercury Analysis in Hydrocarbons
– The Challenges
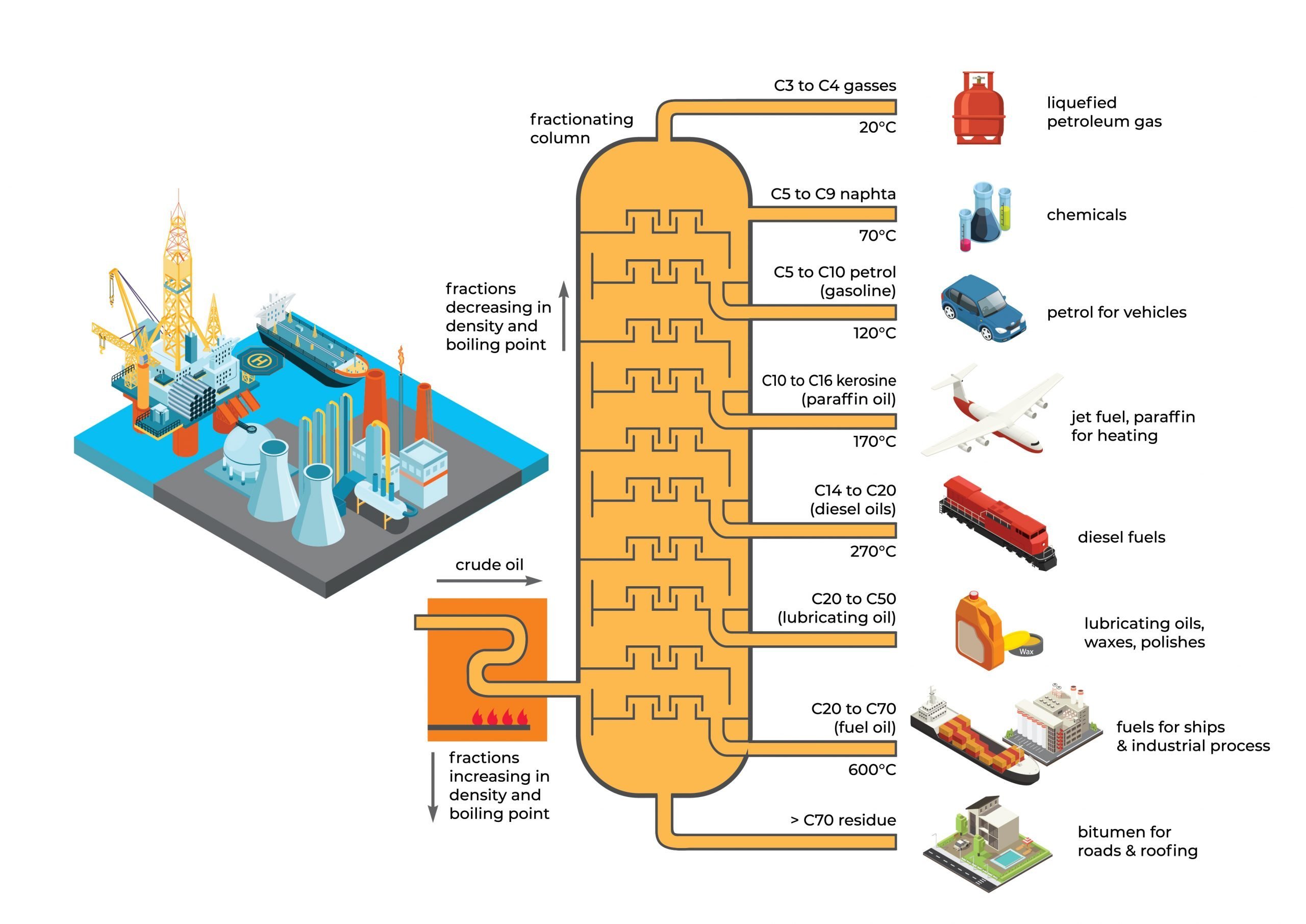
Hydrocarbon is an organic compound consisting of hydrogen and carbon. Liquid hydrocarbons commonly refer to crude oil and its distilled petroleum fractions such as naphtha, gasoline, kerosene and others. Gas hydrocarbon refers to natural gas (i.e. methane) and its refined gases such as propane and butane.
Crude oil is a naturally occurring yellowish-black liquid found in geological formations beneath the Earth’s surface. It is an unrefined petroleum product, which is usually explored, extracted and transported to a refinery to produce usable products such as propane, naphtha, gasoline, diesel, and various other forms of petrochemicals. Natural gas, which is natural occurring and present in abundance, is widely explored for its commercial usage and value. With technology such as GTL (Gas-To-Liquid), natural gas can be synthesized with catalysts into refined products like ethane, propane and butane or liquid blends for automotive fuels.
Mercury is a naturally occurring element, present in virtually all oil and gas, regardless as raw materials or refined products. Mercury levels in crude oil and gas can vary widely, both between and within reservoirs (wells) and geographical regions. Concentrations can be from low ppb (parts per billion) to low ppm (parts per million) levels.
Mercury is not a component in oil and gas, but a contaminant that not only affects the trading value of the oil and gas products but also implicates detrimental effects to the processing plant through amalgamation on metal surfaces and materials, equipment, poisoning of catalysts, and liquid metal embrittlement (LME). LME induces cracking, especially with aluminum, all causing efficiency/downtime in production. Therefore, in the oil and gas industries, mercury is one of the critical measuring parameters on its feeds (crude oil or natural gas) to its products like propane, butane, naphtha, gasoline jet fuel and more.
Liquid hydrocarbons are highly flammable, varying widely in viscosity, flashing point, boiling point, specific gravity, etc. Unlike other heavy metals, mercury has a high vapor pressure making it highly volatile. It presents an analytical challenge to analyze mercury in different liquid hydrocarbon products.
Crude oil contains mostly heavy hydrocarbon chains. Thus, to analyze mercury in crude oil, the analyzer must be able to break down all components, extracting only mercury from it. Therefore, a precise and high enough temperature (typically 800°C or more) control is crucial to decompose all heavy hydrocarbons to ensure complete combustion, turning all hydrocarbons into CO2 and H2O. If the temperature is insufficient, incomplete combustion tends to occur, forming carbon soot that accumulates within the analyzer over time — eventually, causing retention of mercury, memory-effect, and inconsistency in the analysis results.
Naphtha, the lightest liquid fraction refined from crude oil distillation, is also one of the highly prized and in-demand products as feedstock to the petrochemical industry. Naphtha is mainly composed of mixtures with 5~12 carbons, with the boiling point for light naphtha ranging from 35~130℃, during the boiling point of 130~220℃ for the heavier and aromatic naphtha. Naphtha is typically used as feedstock for pyrolysis (cracking) to produce ethylene, propylene, butadiene, benzene, toluene and xylene. These products are, in turn, used to make a wide range of commercial and consumer products like synthetic resins, synthetic rubber, synthetic fiber, dye, medicine and more.
The petrochemical industry normally controls the mercury content in the feedstocks to as low as single-digit parts-per-billion (ppb), preventing and avoiding any unforeseen detrimental effects to their process. Analysis of highly volatile and flammable naphtha is a technical challenge, especially since the mercury content is in the trace level of low parts-per-billion (ppb). Ensuring sample integrity is extremely crucial. One has to prevent loss of analytes (including mercury) from its volatility, controlling the combustion due to its flammability, all to analyze the sample in a quick and precise manner to ensure minimal error. Automated direct syringe injection has proven to be the most reliable.

Solutions
RA-Series
For raw water, process water, industrial wastewater, rain-runoff, seawater, acid digestion (sludges, soil, etc.)

